top of page
Medical Device Blog
Medical Device Regulations ensure safe, effective, and innovative healthcare tools by governing their design, manufacturing, and marketing
Search


Navigating the Canadian Regulation Framework for Medical Devices - AF Pharma's Expert
AF Pharma's team of regulatory experts can guide manufacturers through the entire process, from document preparation to submission.
Soledad


EU Representative - Clinical Trials
According to Article 74 of the CTR, if a sponsor of a Clinical Trial is not based in the EU, appoint a representative is required
Soledad


Affixing a CE Mark in your devices for EU
Some products require CE marking before they can be sold in Europe, like medical devices and machinery products.
Soledad
Do you want to have a CE mark? Follow the steps below
A CE mark logo is an essential identification for certain products in the EU, including medical devices and machinery.
Soledad


Regulating medical devices in the UK - April update
What you need to do to place a medical device on the Great Britain, Northern Ireland and European Union (EU) markets. This is an extract...
Soledad


New FDA Guidance on Risk Management!
Risk management principles are effectively utilized in many areas of business and government including finance, insurance, occupational...
Soledad


Examples of Class III medical devices
Medical Device classification under MDR. Examples of MDR class III medical devices
Soledad


Best Practice Guidance for the Submission of EU Technical File and Documentation of Medical Devices
New guidance on EU Technical Documentation for medical devices
Soledad


Examples of Class IIb medical devices
Medical device classification examples. Class IIb under MDR rules.
Soledad


Examples of Class IIa medical devices
Medical device Class IIa examples
Soledad


Examples of EU Medical Device - Class I
Examples of medical devices classes
Soledad


Impact of extension of the MDR
The European Parliament has voted to adopt an extension of the transition period for the EU Medical Device Regulations and to extend the...
pharmaserviceinc


Foods for special medical purposes/medical foods: A global regulatory synopsis
Foods for special medical purposes/medicinal foods have different regulations depending on where they are manufactured or sold.
Soledad


MDR and IVDR Delay!
Today, 6th January 2023, the Commission adopted a proposal to give more time to certify medical devices to mitigate the risk of...
Soledad


EU Health Commissioner proposes MDR delay to prevent medical device shortages
The European Union Health Commissioner has proposed delaying enforcement of the Medical Devices Regulation (MDR) by three to four years
Soledad


Border line products?
#Manual on #borderline and #classification for #medicaldevices under Regulation (EU) 2017/745 on medical devices and Regulation (EU)...
Soledad


UKCA Marking - extension of standstill period
The MHRA explained its intention to extend the standstill period of using the CE Mark by 12 months to 30 June 2024 for the medical devices.
Soledad

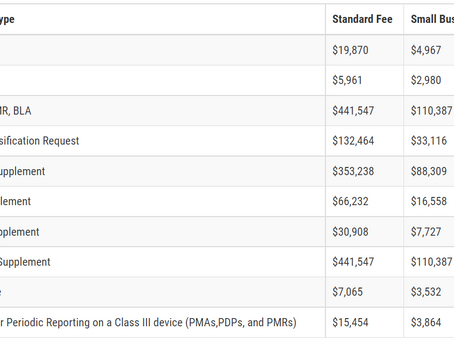
Medical Device User Fee Amendments (MDUFA)
User Fees for FY2023 Annual Establishment Registration Fee: $6,493 All establishments must pay the establishment registration fee. There...
Soledad


FDA NEWS: Electronic Submission Template for Medical Device 510(k) Submissions
This guidance describes the technical standards associated with preparation of the electronic submission template for 510(k)s.
Soledad


UPDATED GUIDANCE MHRA
This guidance document replaces the previous MHRA guidance titled “medical device standalone
software, including apps”.
-
bottom of page

