top of page
Medical Device Blog
Medical Device Regulations ensure safe, effective, and innovative healthcare tools by governing their design, manufacturing, and marketing
Search


FDA allows access to over-the-counter hearing aids
FDA allows access to over-the-counter hearing aids
Soledad


Medical Device Regulation for non-medical products
Medical devices without an intended medical purpose (non-medical products) that fall under the Medical Device Regulation 2017/745.
Soledad


Guidance on Classification Rules for in vitro Diagnostic Medical Devices under Regulation (EU) 2017/
Classification of in vitro diagnostic as per Regulation (EU) 2017/746
Soledad


Diagnostics consultation procedure to the European Medicines Agency - When IVD gets involved
Diagnostics consultation procedure to the European Medicines Agency - When IVD gets involved - companion diagnostics consultation
Soledad

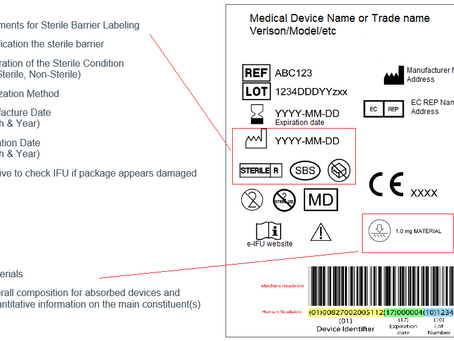
Medical Device Labelling Requirements
A label represents the written, printed or graphic information appearing either on the medical device itself, or on the packaging of each...
pharmaserviceinc


MDCG 2022-9 Summary of safety and performance Template
Regulation (EU) 2017/746 on in vitro diagnostic medical devices (the IVDR) requires that the manufacturer shall draw up a summary of...
pharmaserviceinc


MDSAP AUDIT APPROACH
Maybe you are looking to be into the MDSAP program and to benefit for it. We want to provide you the latest guidance on the MDSAP Audit...
pharmaserviceinc


Guidance on borderline between medical devices and medicinal products under Regulation (EU) 2017/745
Check the final guidance from the MDCG. This document provides further explanations and examples clarifying these provisions in order to...
pharmaserviceinc


EU-TURKEY CUSTOMS UNION AGREEMENT IN THE FIELD OF MEDICAL DEVICES
“Whenever the EU instrument listed above [i.e. Regulations 2017/745 or 2017/746] and the relevant Turkish provisions giving effect to...
pharmaserviceinc


REGULATION (EU) 2022/112 - Updates to the In Vitro Diagnostic Medical Devices Regulation
The amendments made to the IVDR transitional provisions do not postpone the IVDR’s entry into application on 26 May 2022. This is...
pharmaserviceinc


The little updating of notified body under IVDR, judges its implementation
The European Commission's update reveals a growing pipeline of submissions approaching MDR designation, but little hope for a short-term...
pharmaserviceinc


What a United Kingdom Responsible Person (UKRP) does?
After December 31, 2020, after the United Kingdom left the European Union, the United Kingdom faced a new regulatory situation. It has...
pharmaserviceinc


CONCLUSIONS FOR A CLINICAL EVALUATION - MDR
A successful clinical investigation for Medical Device Regulation EU 2017/745 (MDR) is one that generates scientifically valid clinical...
pharmaserviceinc


Declaration of conformity for medical devices
The Declaration of Conformity (also known as DoC) is a critical document for every medical device in Europe.
pharmaserviceinc


Are you looking for an MDR Technical File template?
MDR Technical File template
pharmaserviceinc


Content of Premarket Submissions for Device Software Functions
The FDA just published draft guidance on Premarket Submissions for Device Software Functions. It describes the information the FDA...
pharmaserviceinc


Formal proposal to delay IVDR
#BreakingNews We now have a formal proposal from the European Commission to delay #IVDR. The proposal does not change any requirements of...
pharmaserviceinc


A new MDCG Guidance has just been released, namely 2021-25
The document addresses a crucial issue, often confusing for manufacturers, authorized representatives, importers and distributors, the...
pharmaserviceinc
bottom of page

